Synthesis of Drugs and Bioactive Compounds
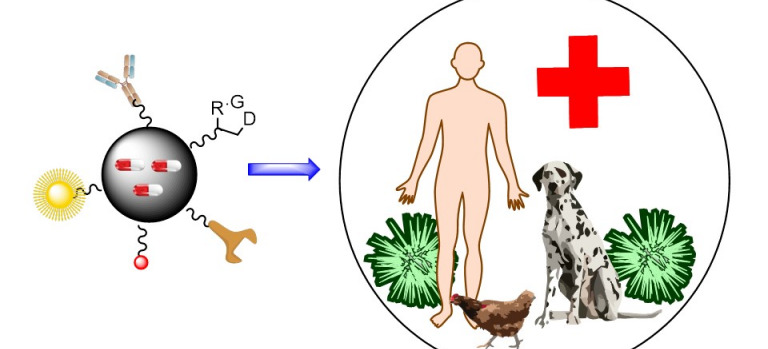
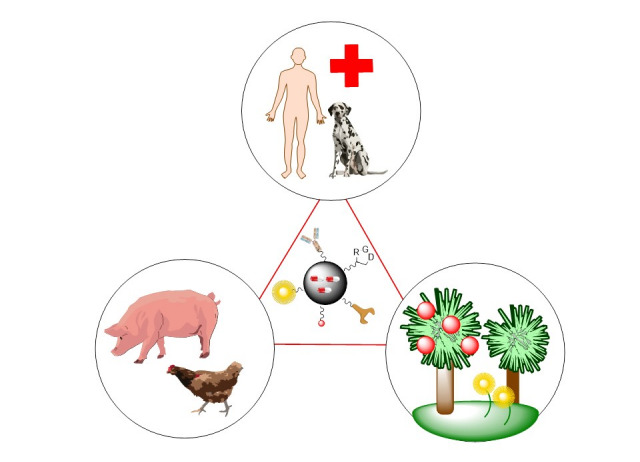
The Synthesis of Drugs and Bioactive Compounds group, created in 2004, is dedicated to the development of new synthetic methodologies and their application to obtain compounds of pharmacological and agricultural interest (One Health).
These compounds include nucleoside analogues, peptides and peptidomimetics (e.g. antimicrobial peptides and quorum inhibitors), fluorescent drug and medical probe derivatives, nanodrugs and crop protection agents.
The Synthesis of Drugs and Bioactive Compounds group profile page on Digital.CSIC.
Presentation
-
Research lines
Development of more efficient chemical processes with less environmental impact
Sequential processes are developed that link various ionic and radical reactions. Intermediates do not need to be isolated and materials, energy and time are saved, and the waste to be treated is reduced. These processes have been used to manufacture a wide variety of bioactive compounds, such...

Site-selective peptide modification to develop drugs and peptide catalysts
Selective peptide modification allows easy generation of varied peptide libraries from a few starting substrates, and saves time and resources in the process of drug and peptide catalyst discovery. Our group has introduced "modifiable units" that, once introduced in peptides, can be easily...
Preparation of antimicrobial peptides and quorum sensing inhibitors
Our group is developing antimicrobial peptides for biomedicine, livestock and agriculture, and also compounds with potential quorum sensing inhibitory action. Several of the results have been patented and have led to international communications.
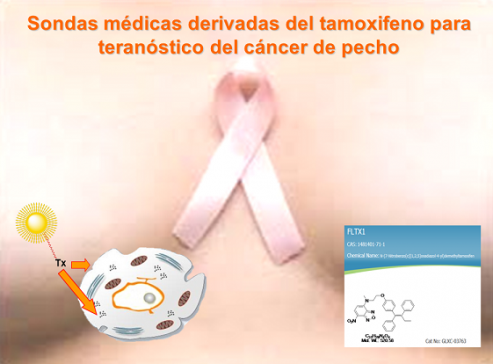
Development of fluorescent drug derivatives and medical probes
In collaboration with ULL, fluorescent analogues of tamoxifen, an anti-tumour agent widely used to treat breast cancer, have been developed as medical probes and new drugs.
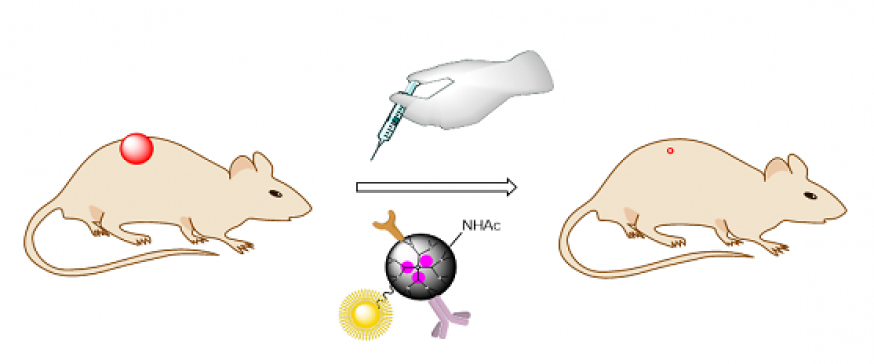
Nanodrug development
In collaboration with several Biotechnology and Nanotechnology groups, we are preparing nano-drugs for use as antimicrobials and antitumor agents, from phototherapy to nanoparticles for selective transport of antitumor agents, antimicrobials or medical probes. The nanoparticles are decorated...

Development of protective agents for crops
In collaboration with two IPNA Agrobiology groups, we are developing crop protection compounds against phytopathogens and abiotic stresses such as drought and salinity.
PhD & MSc. Thesis
Nuevos métodos para convertir sustratos de bajo coste en compuestos de alto valor añadido, por su interés químico y farmacéutico
Esta tesis obtuvo la Mención Industrial y ha sido la primera tesis en la ULL del programa de Doctorados Industriales. Calificación: Sobresaliente cum laude.
Directora: Alicia Boto (CSIC)
Codirectores: Dácil Hernández (CSIC) y Gabriel García-Llanos (BIOSIGMA SL)
Carmen Carro Sabina
Procesos secuenciales para la síntesis de aminas quirales a partir de amino ácidos y de péptidos
Modificación de productos Bioactivos: Síntesis de derivados de aminoácidos, péptidos y fármacos antitumorales
Procesos secuenciales de escisión radicalaria-oxidación-adición de nucleófilos: aplicación a la modificación selectiva de péptidos y a la síntesis de productos bioactivos
Síntesis estereoselectiva de análogos de alcaloides y de nucleósidos por medio de un proceso secuencial de fragmentación-adición de nucleófilos
Modificación de productos Bioactivos: Aproximación a la Síntesis de análogos simplificados de enzimas
Procesos Secuenciales estereoselectivos para la síntesis de precursores de productos bioactivos
Síntesis en un paso de beta-amino ácidos a partir de alfa-amino ácidos. Preparación de alfa,beta-péptidos
Reactividad de Radicales Alcoxilo y Carboxilo. Abstracción Intramolecular de Hidrógeno y beta-Fragmentación: Aplicaciones Sintéticas
Estudio del Mecanismo de Fragmentación de Radicales Alcoxilo Anoméricos y Radicales Carboxilo: Intermedios Radicalarios e Iónicos. Aplicaciones a la Síntesis de Carbociclos y Heterociclos Nitrogenados
Funding

Nuevos agentes para la lucha selectiva contra patógenos: Inhibidores del Quorum Sensing y péptidos antimicrobianos patógenoespecíficos (SELECTFIGHT)
En Ejecución

Preparacion de peptidos antimicrobianos de uso en salud humana y agricultura (SAF2013-48399-R)
Finalizado

Preparación de péptidos antimicrobianos de uso en salud humana y agricultura
Finalizado

Preparación de péptidos antimicrobianos de uso en salud humana y agricultura
Finalizado

Utilización de los desechos del cultivo del plátano de Canarias: Aplicabilidad frente a patologías de la piel
Raquel Marín (Universidad de La Laguna)
Finalizado

Métodos prácticos, de alcance y bajo coste para despertar vocaciones científicas en épocas de crisis (FCT-14-8265)
Finalizado

Drug discovery for the treatment of selected protozoa (Toxoplasma, Neospora and Acanthamoeba)
Hany Elsheikha (Universidad de Nottingham)
Finalizado

Nuevas metodologías para la modificación selectiva de péptidos (bioactivos o catalíticos) y para obtener azanucleotidos (CTQ2009-07109)
Finalizado

Síntesis estereoselectiva de análogos de alcaloides citotóxicos
Finalizado

Síntesis Estereoselectiva de Compuestos Nitrogenados con Posible Actividad Farmacológica: Amino ácidos, Azanucleósidos, Alcaloides y Análogos Sintéticos (CTQ2006-14260)
Finalizado

Síntesis de Alcaloides Bioactivos y Análogos Sintéticos a partir de Amino Ácidos
Finalizado

Síntesis de Alcaloides y Nucleósidos Acíclicos con Potencial Actividad Farmacológica a partir de Amino Ácidos y Carbohidratos
Rosendo Hernández González
Finalizado

Nueva Metodología Sintética para la Obtención de Alcaloides con Actividad Biológica y Análogos Sintéticos a partir de Aminoácidos
Rosendo Hernández González
Finalizado
People
Publications
Conversion of "customizable Units" into N-Alkyl Amino Acids and Generation of N-Alkyl Peptides
An efficient conversion of hydroxyproline >customizable> units into new amino acids with a variety of N-alkyl substituents is described. The process is versatile and can afford valuable N-methyl amino acids and N,O-acetals. In addition, it allows the introduction of N-homoallylic substituents and N-chains with terminal ester, ketone, or cyano groups. These chains could be used for peptide extension or conjugation to other molecules (e.g., by olefin metathesis, peptide ligation, etc.). The transformation is carried out in just two (for R = CHOAc) or three steps (scission of the pyrrolidine ring, manipulation of the α-chain, and the N-substituent) under mild, metal-free conditions, affording products with high optical purity.
Saavedra, Carlos J.; Carro, Carmen; Hernández, Dácil; Boto, Alicia
Metal-Free, Site-Selective Peptide Modification by Conversion of “Customizable” Units into β-Substituted Dehydroamino Acids
Our site-selective modification of serine or threonine units in peptides allows the generation of β-substituted dehydroamino acids, which increase peptide resistance to hydrolysis and may improve their biological properties. Both the terminal and internal positions can be modified, and different customizable units can be activated separately. Remarkably, high Z selectivity is achieved, even at internal positions. The conversion involves a one-pot oxidative radical scission/phosphorylation process by using the low-toxicity (diacetoxyiodo)benzene/iodine system as the scission reagent. The resulting α-amino phosphonates undergo a Horner-Wadsworth-Emmons reaction to produce the dehydroamino acid derivatives (in a Z/E ratio of usually >98:2) under mild and metal-free conditions.
Saavedra, Carlos Javier; Hernández, Dácil; Boto, Alicia
Domino Process Achieves Site-Selective Peptide Modification with High Optical Purity. Applications to Chain Diversification and Peptide Ligation
The development of peptide libraries by site-selective modification of a few parent peptides would save valuable time and materials in discovery processes but still is a difficult synthetic challenge. Herein, we introduce natural hydroxyproline as a convertible unit for the production of a variety of optically pure amino acids, including expensive N-alkyl amino acids, homoserine lactones, and Agl lactams, and to achieve the mild, efficient, and site-selective modification of peptides. A domino process is used to cleave the customizable Hyp unit under mild, metal-free conditions. Both terminal and internal positions can be modified, and similar customizable units can be differentiated. The resulting products possess two reactive chains which can be manipulated independently. The versatility and scope of this process is highlighted by its application to the ligation of two peptide chains, and the generation of peptides with several chains and peptides with conformational restrictions.
Romero Estudillo, Iván Omar; Boto, Alicia
Nucleoside analogues: Synthesis and biological properties of azanucleoside derivatives
The azanucleosides are nucleoside analogues where the furanose ring is replaced by a nitrogen‐containing ring or chain. Many azanucleosides are potent antiviral, anticancer and antimicrobial agents, or serve as valuable components of oligonucleotides with improved stability, binding or hybridization properties. Therefore, the development of new analogues is a very active area in medicinal and synthetic chemistry. Their synthesis and their interesting biological properties are discussed in this microreview.
Hernández, Dácil; Boto, Alicia
Unique SERM-like properties of the novel fluorescent tamoxifen derivative FLTX1
Tamoxifen is a selective estrogen receptor modulator extensively used on estrogen receptor-positive breast cancer treatment. However, clinical evidences demonstrate the increased incidence of undesirable side effects during chronic therapies, the most life threatening being uterine cancers. Some of these effects are related to tissue-dependent estrogenic actions of tamoxifen, but the exact mechanisms remain poorly understood. We have designed and synthesized a novel fluorescent tamoxifen derivative, FLTX1, and characterized its biological and pharmacological activities. Using confocal microscopy, we demonstrate that FLTX1 colocalizes with estrogen receptor α (ERα). Competition studies showed that FLTX1 binding was totally displaced by unlabeled tamoxifen and partially by estradiol, indicating the existence of non-ER-related triphenylethylene-binding sites. Ligand binding assays showed that FLTX1 exhibits similar affinity for ER than tamoxifen. FLTX1 exhibited antiestrogenic activity comparable to tamoxifen in MCF7 and T47D cells transfected with 3xERE-luciferase reporter. Interestingly, FLTX1 lacked the strong agonistic effect of tamoxifen on ERα-dependent transcriptional activity. Additionally, in vivo assays in mice revealed that unlike tamoxifen, FLTX1 was devoid of estrogenic uterotrophic effects, lacked of hyperplasic and hypertrophic effects, and failed to alter basal proliferating cell nuclear antigen immunoreactivity. In the rat uterine model of estrogenicity/antiestrogenicity, FLTX1 displayed antagonistic activity comparable to tamoxifen at lower doses, and only estrogenic uterotrophy at the highest dose. We conclude that the fluorescent derivative FLTX1 is not only a suitable probe for studies on the molecular pharmacology of tamoxifen, but also a potential therapeutic substitute to tamoxifen, endowed with potent antiestrogenic properties but devoid of uterine estrogenicity.
Marrero-Alonso, Jorge; Morales, Araceli; García Marrero, B.; Boto, Alicia; Marín, Raquel; Cury, Débora; Gómez, Tomás; Fernández-Pérez, Leandro; Lahoz Zamarro, Fernando; Díaz, Mario
Conformation and Chiral Effects in α,β,α-Tripeptides
Short α,β,α-tripeptides comprising a central chiral trisubstituted β2,2,3*-amino acid residue form unusual γ-turns and δ-turns in CDCl3 and DMSO-d6 solutions but do not form β-turns. Thermal coefficients of backbone amide protons, 2D-NMR spectra, and molecular modeling revealed that these motifs were strongly dependent on the configuration (chiral effect) of the central β-amino acid residue within the triad. Accordingly, SSS tripeptides adopted an intraresidual γ-turn like (C6) arrangement in the central β-amino acid, whereas SRS diastereomers preferred an extended δ-turn (C9) conformation. A different SRS-stabilizing bias was observed in the crystal structures of the same compounds, which shared the extended δ-turn (C9) found in solution, but incorporated an additional extended β-turn (C11) to form an overlapped double turn motif.
Saavedra, Carlos J.; Boto, Alicia; Hernández, Rosendo; Miranda, José Ignacio; Aizpurua, Jesus M.
Collaborations
Carlos Javier Saavedra Fernández
Universidad de La Laguna
E-mail: csaavedr [at] ull.edu.es
Fernando Lobo Palacios
Universidad de La Laguna
Alicia Boto Castro
Contact information
Galería de imágenes y vídeos
News/Blog
- 19 January 2021
- 22 July 2020
- 31 January 2020
- 25 April 2019
Other research groups