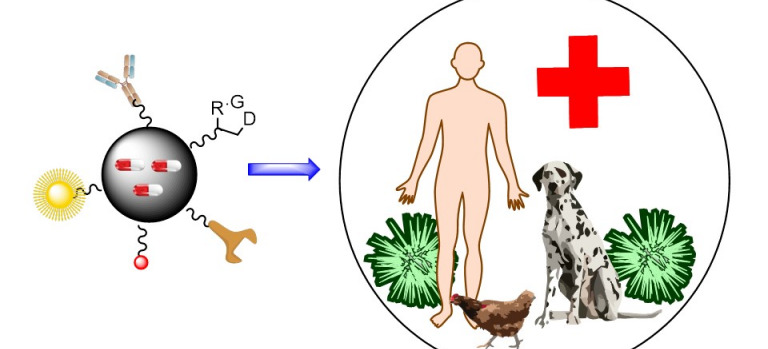
Natural Products Synthesis
The Natural Products Synthesis Group (SIPN), one of the oldest in the IPNA-CSIC, seeks to develop new synthetic methodologies based on the use of light, for selective modification of biomolecules to obtain products of interest.
The Natural Products Synthesis Group profile page on Digital.CSIC.
Presentation
The research lines of our group have evolved to focus on the field of synthetic methodology, especially radical chemistry. Thus, the reactions of β-fragmentation (β-FRA) and intramolecular transfer of hydrogen (TIH) promoted by alcoxyl or amidyl radicals, in the presence of hypervalent iodine reagents, such as (diacetoxyiodine)benzene (DIB) or iodine-sylbenzene, and iodine with excellent results, have been extensively studied, fine-tuning a methodology, known as the Suarez Reaction, which internationally characterises our group. This protocol involves the regioselective functionalization of C-H links in a highly efficient and sustainable manner.
Among the most interesting novel contributions of the Suarez conditions are their application to carbohydrates and steroids to obtain a wide battery of glycomimetics and chiral synthetic forms, which are difficult to obtain by other methods.
Currently, we have extended this methodology to other types of accessible natural enantiopure products. Additionally, other processes for the chemoselective modification of organic molecules are being studied under diverse reaction conditions, such as in reducing medium, photochemicals or other applications of the various redox systems of halogens.
Research lines
Oxidative and photoredox decarboxylation of cyclodextrins
This line of research focuses on the study of the radical β-fragmentation reaction induced by light to promote the decarboxylation of mono- and di-carboxylic acids in cyclic polysaccharide systems, such as cyclodextrins.
Intramolecular Hydrogen Atom Transfer Reactions (HAT) promoted by photochemical excited carbonyls
This research line started 10 years ago and is focused on the study of HAT reactions promoted by photochemically excited carbonyls.
Intramolecular Transfer Processes 1,8 in Complex Carbohydrate Systems
Intramolecular hydrogen transfer (IHT) radical reactions involving six- and seven-member transition states are very common, while those via seven-member transition states are scarce. Recently, our group has pioneered an unusual 1,8-TIH process in (1→4)-O-disaccharides systems promoted by the 6-O...
Alkoxyl Radical Fragmentation Reaction
The alkoxyl radical fragmentation (ARF) reaction1 is a research line that has been developed by this laboratory. In previous studies2 by our research group, we have described that starting from carbohydrates (a) by β-fragmentation of alkoxyl radicals (FRA), generated by...
GeoBiomimetic Applications of the Iodine Reactivity in the Field of the Organic Synthesis
In this research line, our efforts are focused on a better understanding on the fascinating redox iodine systems in different environments. A relevant observed feature of this redox system has been the dynamicity within the iodine transient species interconversions driving us to the concept of...
PhD & MSc. Thesis
Síntesis de Compuestos Organofosforados Altamente Funcionalizados, mediante Fragmentación de Radicales Alcoxilo Anóméricos de Carbohidratos. Aplicaciones Sintéticas
Desarrollo de nuevas metodologías para la síntesis de azepanos
Desarrollo de Nuevas Metodologías para la síntesis de inhibidores de glicosidasas y glicosiltransferasas y aplicación de la fotociclación de 1,2-dicetonas en el diseño de nuevos derivados polihidroxilados
Reacciones de Transferencia de Hidrógeno en Sistemas de Furanosas
Transferencia de átomos de Hidrógeno promovida por excitación fotoquímica de 1,2-dicetonas
TFG | Efectos del ácido glutámico y tres derivados en respuesta a estrés hídrico aplicado sobre Vicia faba L.
TFG | Efecto del ácido glutámico y su tosil-derivado en el crecimiento y respuesta al déficit hídrico en judía (Vicia faba L.)
TESIS | Funcionalización Selectiva de Enlaces C(sp3)–H Alifáticos usando Amidas como Grupos Directores y Sistemas Reactivos Basados en Yodo Hipervalente/yodo: Formación de Enlaces C-N Mediante Transferencia Intramolecular de Átomos de Hidrógeno
Supervisión de contratos en prácticas
Dirección de Estudiantes Universitarios Extranjeros
Dirección de prácticas de Fin de Ciclo Formativo de Técnico de Grado Superior de Química Ambiental
MÁSTER | Nuevos Sustituyentes sobre Radicales Centrados en Nitrógeno. Estudio de su Reactividad y Aplicaciones en Síntesis Asimétrica
TESIS | Fragmentación radicalaria de alcoholes anoméricos de carbohidratos: Síntesis de Heterociclos Polihidroxilados y Evaluación de su Actividad Biológica
MÁSTER | Desarrollo de Nuevas Metodologías para la Síntesis de Azepanos
Funding

Oler para creer | Actividades paralelas
Desarrollo y ejecución de las actividades paralelas vinculadas a la exposición 'Oler para creer. La química del olor'.
Inés Pérez Martín

Estudios mecanísticos y aplicaciones metodológicas de la N-glicosilación de guanidinas: el caso de la ramnosilación de argininas por la enzima bacteriana EarP (N-RHAGED)
Proyecto bajo la gestión de la Agencia Española de Investigación. Este proyecto se enmarca en el Programa Estatal para Impulsar la Investigación Científico-Técnica y su Transferencia, del Plan…
En Ejecución

Oler para creer. La química del olor
'Oler para creer. La química del olor' es una exposición que propone entrelazar ciencia, historia y tecnología para ofrecer experiencias olfativas que conecten con los visitantes de una forma…
En Ejecución

El volcán de Cumbre Vieja por tierra, mar y aire
El proyecto 'El volcán de Cumbre Vieja por tierra, mar y aire' se materializó en la exposición 'Ceniza y lava. Revelaciones científicas junto al volcán', una experiencia inmersiva que invitaba a…
Finalizado

Estudio exhaustivo de las defensas inducidas en plantas mediante la aplicación exógena de derivados fluorescentes de MSB, vitamina K3 y bisulfito sódico (VK3-BODIPY)
Finalizado

Radicales Libres en la Síntesis de Nuevos Glicomiméticos con Potencial Actividad Terapeútica
Finalizado

Preparación de Nuevas Estructuras de Ciclodextrinas para el Transporte de Fármacos
Finalizado

Aplicaciones Sintéticas de los Procesos de Transferencia Intramolecular de Hidrógeno en Sistemas de Carbohidratos. Obtención de Nuevos Sintones Quirales
Finalizado

Diseño de compuestos enantioméricamente puros utilizando nuevas metodologias basadas en C-, N-y O-radicales. Aplicación a la síntesis de sustancias con actividad biológica
Finalizado

Reacciones de Transferencia Intramolecular de Hidrógeno sobre Carbohidratos. Nuevas Metodologías para la Síntesis de Compuestos Potencialmente Bioactivos
Finalizado
People
Elisa Isabel De León Alonso
María del Sol Rodríguez Morales
Concepción González Martín
Antonio Jesús Herrera González
María Ángeles Martín Hernández
Inés Pérez Martín
Esther María Martínez González
Nieves Rodríguez Paz
Andrés González Santana
Ricardo Guillermo Álvarez
Publications
Synthesis of Chiral Polyhydroxylated Benzimidazoles by a Tandem Radical Fragmentation/Cyclization Reaction: A Straight Avenue to Fused Aromatic-Carbohydrate Hybrids
The synthesis of benzimidazole-fused iminosugars through a tandem β-fragmentation-intramolecular cyclization reaction is described. The use of the benzimidazole ring as the internal nucleophile and the use of phenyliodosophthalate (PhI(Phth)), a new metal-free and low toxic hypervalent iodine reagent, are the most remarkable novelties of this synthetic strategy. With this approach, we have demonstrated the usefulness of the fragmentation of anomeric alkoxyl radicals promoted by the PhI(Phth)/I system for the preparation of new compounds with potential interest for both medicinal and synthetic chemists.
André-Joyaux, Emy; Santana, Andrés G.; González Martín, Concepción C.
Reductive Radical Cascades Triggered by Alkoxyl Radicals in the β-Cyclodextrin Framework
The generation and fate of 2,3,6-icosa-O-methyl-β-cyclomaltoheptaos-6-O-yl radical under reductive conditions is described. Two radical cascade reactions are involved: the main one is triggered by a 1,8-HAT of the hydrogen at 5C. The radical can reach the anomeric hydrogen at 1C three sugar units ahead using a six-step sequence. The different hydrogen donor ability of the group 14 hydrides permits one to selectively stop the cascade at 5C, 2C, and 4C to obtain β-CD with a β-l-Idop unit, acyclic hepta-, and hexa-saccharide structures, respectively.
León, Elisa I.; Martín, Ángeles; Pérez-Martín, Inés; Suárez, Ernesto
Chemoselective Intramolecular Functionalization of Methyl Groups in Nonconstrained Molecules Promoted by N-Iodosulfonamides
Mechanistic evidence observed in Hofmann–Löffler–Freytag-type reactions has been crucial to achieve the chemoselective functionalization of methyl groups under mild conditions. Radical-mediated methyl iodination and subsequent oxidative deiodination are the key steps in this functionalization, where iodine chemistry has a pivotal role on the formation of the C–N bond. The concepts of single hydrogen atom transfer (SHAT) and multiple hydrogen atom transfer (MHAT) are introduced to describe the observed chemoselectivity.
Paz, Nieves R.; Rodríguez Sosa, Dionisio; Valdés, Haydée; Marticorena, Ricardo; Melián, Daniel; Copano, Belén; González Martín, Concepción C.; Herrera, Antonio J.
Easy access to modified cyclodextrins by an intramolecular radical approach
A simple method to modify the primary face of cyclodextrins (CDs) is described. The 6I‐O‐yl radical of α‐, β‐, and γ‐CDs regioselectively abstracts the H5II, located in the adjacent D‐glucose unit, by an intramolecular 1,8‐hydrogen‐atom‐transfer reaction through a geometrically restricted nine‐membered transition state to give a stable 1,3,5‐trioxocane ring. The reaction has been extended to the 1,4‐diols of α‐ and β‐CD to give the corresponding bis(trioxocane)s. The C2‐symmetric bis(trioxocane) corresponding to the α‐CD is a stable crystalline solid whose structure was confirmed by X‐ray diffraction analysis. The calculated geometric parameters confirm that the primary face is severely distorted toward a narrower elliptical shape for this rim.
Álvarez-Dorta, Dimitri; León, Elisa I.; Kennedy, Alan R.; Martín, Ángeles; Pérez-Martín, Inés; Suárez, Ernesto
Fragmentation of carbohydrate anomeric alkoxyl radicals: Synthesis of chiral polyhydroxylated β-iodo- and alkenylorganophosphorus(V) compounds
A direct approach to β‐iodophosphonates and β‐iodophosphine oxides from 2,3‐dideoxy‐3‐phosphoryl carbohydrate derivatives has been achieved by using the anomeric alkoxyl radical 1,2‐fragmentation protocol. The reaction has been conducted on carbohydrate derivatives under mild conditions with (diacetoxyiodo)benzene and molecular iodine. Subsequent dehydroiodination afforded the corresponding vinylphosphonates and vinylphosphine oxides.
Hernández-Guerra, Daniel; Rodríguez Morales, María S.; Suárez, Ernesto
Sequential Norrish type II photoelimination and intramolecular aldol cyclization of α-diketones: Synthesis of polyhydroxylated cyclopentitols by ring contraction of hexopyranose carbohydrate derivatives
The excitation of the innermost carbonyl of nono‐2,3‐diulose derivatives by irradiation with visible‐light initiates a sequential Norrish type II photoelimination and aldol cyclization process that finally gives polyfunctionalized cyclopentitols. The rearrangement has been confirmed by the isolation of stable acyclic photoenol intermediates that can be independently cyclized by a thermal 5‐(enolexo)‐exo‐trig uncatalyzed aldol reaction with high diastereoselectivity. In this last step, the large deuterium kinetic isotope effect found for the 1,5‐hydrogen atom transfer seems to indicate that the aldol reaction runs through a concerted pericyclic mechanism. Owing to the ready availability of pyranose sugars of various configurations, this protocol has been used to study the influence of pyranose ring‐substituents on the diastereoselectivity of the aldol cyclization reaction. In contrast with other pyranose ring contraction methodologies no transition‐metal reagents are needed and the sequential rearrangement occurs simply by using visible light and moderate heating (0 to 60 °C).
Álvarez‐Dorta, Dimitri; León, Elisa I.; Kennedy, Alan R.; Martín, Ángeles; Pérez-Martín, Inés; Riesco-Fagundo, Concepción; Suárez, Ernesto
Concepción González Martín

Contact information
News/Blog
- 20 August 2019
Other research groups