Enantiodivergent cyclization by inversion of the reactivity in ambiphilic molecules
One of the big problems for pharmaceutical and chemical companies is the need to have both enantiomers of a particular chiral compound when testing its efficacy or toxicity. Enantiomers are stereoisomers whose molecular structures are mirror images that cannot be superimposed on each other, so they do not match in all parts and may therefore have differences in their chemical properties when they interact with other chiral molecules. For this reason, they present very different biological activities, since most of the molecules present in living beings are chiral.
This situation has driven the development of a large number of enantiodivergent processes from achiral substrates, an approach that requires the use of both enantiomers or pseudoenantiomers of a reagent that can act as a binder or as a catalyst. However, this approach has a serious drawback, and that is that most natural chiral molecules are accessible in only one absolute configuration. Another interesting alternative way of obtaining both enantiomers is the enantiodivergent strategy, which uses chiral substrates as starting products. In this case, two main approaches can be presented: one sequentially modifies the functional groups, without altering the stereochemistry of the chiral elements present in the initial substrate, and the other implies changes in the stereochemistry, which can be defined as an enantiodivergent reaction. In both cases, only one enantiomer of the starting material is required, and the use of chiral reagents or catalysts is optional.
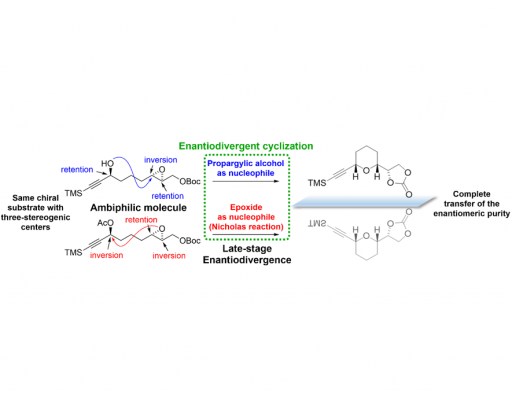
There are very few examples of enantiodivergent reactions, and they generally occur with a single element of symmetry, such as a stereogenic centre or a chiral axis. For this reason, it is a great challenge for synthetic organic chemists to find enantiodivergent reactions where a larger number of stereogenic centres are involved. The advantage of this approach is that it makes it possible to obtain both enantiomers at a more advanced stage of synthesis. Our idea is based on inverting the reactivity of functional groups present in an ambiphilic molecule (a molecule carrying both electrophilic and nucleophilic sites).
As a test of this concept, we focused on the synthesis of cyclic ethers, and considered that an epoxy and a proparyl alcohol placed in appropriate positions in a linear precursor are suitable functional groups for this objective. Thus, one could act as a nucleophile and the other as an electrophile or vice versa, providing an enantiodivergent route to obtain both enantiomers of the final cyclic ether. In this paper we present the first example of an enantiodivergence process achieved by reversing the reactivity in ambiphilic molecules, and it is also the first time that an enantiodivergent reaction encompasses more than one stereogenic centre.
In summary, we have introduced the concept of enantiodivergence by reversing the reactivity of specific functional groups in ambiphilic precursors. This opens a new approach to the synthesis of any of the enantiomers of a compound from a common chiral substrate. It also allows enantiodivergence to be performed at an advanced stage of synthesis. There is thus a greater potential to reduce the synthetic effort when both enantiomers of a given compound are required.
To learn more, check the article here: "Enantiodivergent cyclization by inversion of the reactivity in ambiphilic molecules".
By Tomás Martín Ruíz.