Physiological Traits Combination Shapes Common Strategies of Water and Carbon Use Regulation Across Fruit Tree Species
Crop plants, including fruit trees, are particularly vulnerable to water scarcity because past selection prioritized productivity over drought resistance, making it challenging to maintain productivity with minimal water use in the context of climate change. This study aims to determine which trait combination of 10 fruit tree species influences their water and carbon use, with the goal of understanding their adaptability to water scarcity. The results showed that water stress traits (turgor loss point, TLP; vulnerability index, VI), a carbon-related trait (specific leaf area; SLA), and a biomass allocation trait (Huber value; Hv) define the major axis of variability and present the strongest correlations with other traits. Two distinct strategies emerged: the first, mainly around Prunus species, was characterized by high Hv, low SLA, more negative TLP, and low VI, indicating greater water-stress tolerance due to sapwood redundancy and reduced organ vulnerability. They also exhibited higher maximum photosynthetic rates, indicating greater assimilation rates. The second strategy, mainly including Citrus species, exhibited opposite traits and trends. These trait combinations were likely shaped by shared ancestry and environmental factors. Understanding these correlations can guide irrigation practices and the selection of resilient species, contributing to more robust agricultural systems in a changing climate scenario.
Hernández Santana, V.; Sebastián Azcona, Jaime; Rodríguez Domínguez, Celia M.; Pérez Martín, Alfonso; Montero de Espinosa, Antonio; Benzal Moreno, Daniel; Rossi, Federica; Pérez-Romero, Luis F.; Díaz-Espejo, Antonio.
ABA-receptor agonist iSB09 decreases soil water consumption and increases tomato CO2 assimilation and water use efficiency under drought stress
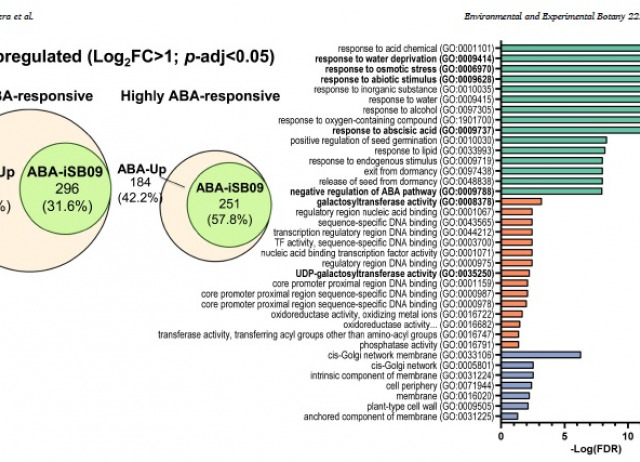
Climate change can alter precipitation patterns, disrupting the natural water cycle and generating drought periods that negatively impact crop yield or plant survival. Novel biotechnological approaches are being developed to face water deficits. Specifically, molecular knowledge of the plant hormone abscisic acid (ABA) can be harnessed to develop genetic and chemical approaches to cope with abiotic stress. ABA receptor agonists are promising molecules that activate ABA signaling on demand and show long-lasting effects, in contrast to the exogenous application of ABA, which has a short half-life. In this work, we studied the effect of the iSB09 agonist on tomato plants grown under drought stress or well-watered conditions. iSB09 treatment induced stomatal closure in tomato through activation of PYL1-like and PYL4-like ABA receptors. Additionally, RNA-seq analyses reveal coordinated upregulation by ABA or iSB09 of the genes encoding enzymes involved in the synthesis of the osmoprotective galactinol and raffinose family of oligosaccharides. Foliar spraying of iSB09 under drought conditions anticipated the regulation of transpiration, promoted drought avoidance and increased water use efficiency in tomato plants. Physiological analysis of agonist-treated plants reveals increased CO2 assimilation and effective quantum yield of the photosystem II under drought conditions in iSB09-treated plants compared to mock-treated. Faster regulation of transpiration at the start of the drought period was achieved by iSB09 treatment, and, as a result, water consumption was reduced compared to mock-treated plants. Overall, the agonist treatment mounts the genome-wide transcriptional response to stress and increases water use efficiency under drought conditions and plant protection.
Sanchez-Olvera, Mayra; Martin-Vasquez, Constanza; Mayordomo, Cristian; Illescas-Miranda, Jonatan; Bono, Mar; Coego González, Alberto; Jana Alonso Lorenzo ; Hernández-González, Mercedes; Jiménez-Arias, David; Forment, Javier; Albert, Armando; Granell, Antonio; Andrés, A. ; Rodriguez, Pedro L.
No detectable truncating mutations in large T antigen (LT-Ag) sequence of Merkel cell polyomavirus (MCPyV) DNA obtained from porocarcinomas
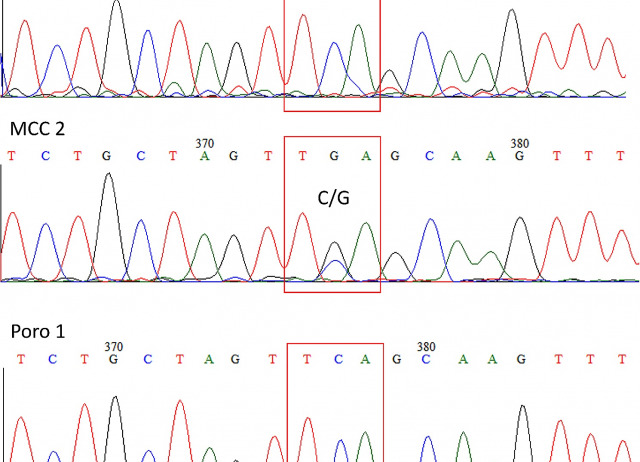
Merkel cell polyomavirus (MCPyV) is associated with Merkel cell carcinoma (MCC). In tumor cells the MCPyV large T antigen (LT-Ag) is frequently found truncated and this is considered a major tumor-specific signature. The role of MCPyV in other, non-MCC tumours, is little known. Viral DNA and/or tumour-specific mutations have been sometimes detected in different tumours, but such data are not unequivocal and the involvement of the virus in the tumorigenesis is not clear. In a previous study, we demonstrated a significantly higher prevalence of MCPyV DNA in formalin fixed paraffin embedded (FFPE) porocarcinoma tissues compared to the normal skin.
In the present study, we investigated the presence of truncating mutations in MCPyV LT-Ag coding region in porocarcinoma specimens. Using several overlapped PCR primer pairs, the complete LT-Ag sequence from two biopsies were obtained. No truncating mutations were detected.
The lack of truncating mutations in LT-Ag sequence does not seem to support the role of MCPyV in porocarcinoma oncogenesis. However, an oncogenetic mechanism, different from that proposed for MCC and not associated with the LT-Ag mutations/deletions, cannot be excluded. Further studies of more sequences coding for LT-Ag would be needed to verify this hypothesis.
Arvia, Rosaria; Sollai, Mauro; Massi, Daniela; Asensio-Calavia, Patricia; Urso, Carmelo; Zakrzewska, Krystyna.
Foliar treatment with MSB (menadione sodium bisulphite) to increase artemisinin content in Artemisia annua plants
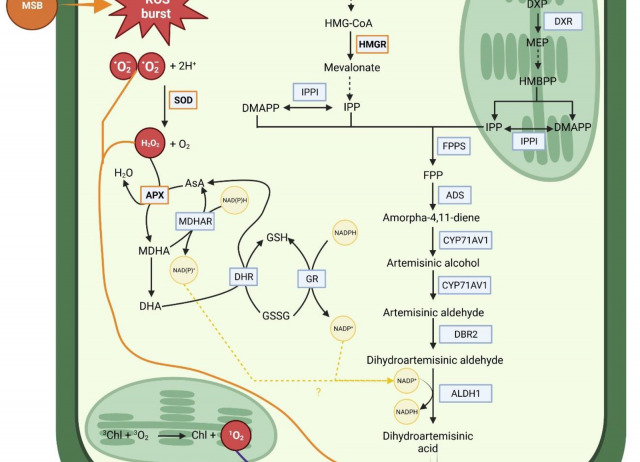
Artemisinin is a sesquiterpene lactone endoperoxide, recognized for its application as a treatment for malaria. Besides, it has activity against several types of cancers, as well as anti-viral and anti-inflammatory properties. The chemical synthesis of artemisinin is quite complex and economically unfeasible. Hence, artemisinin is typically obtained from Artemisia annua plants in low quantities in relation to the biomass. A strategy to increase the biosynthesis of secondary metabolites would be the application of elicitors. In this work, the effect of treating A. annua plants with menadione sodium bisulphite (MSB) is studied. An initial dose-optimization was conducted by treating the plants with different MBS concentrations (0, 0.2, 0.8, 1, 2, 3 and 4 mM) and artemisinin was quantified 48 h after treatment. The highest artemisinin content (3.71 mg artemisinin g−1 DW) was obtained with 1 mM of MSB. Then, harvesting time was optimised (24, 48 and 72 h after treatment application) with 1 mM of MSB. The largest increase in artemisinin content was observed at 48 h with an increment of 62.37 % over the control. The treatment increased H2O2 content, as well as the activity of superoxide dismutase (SOD) and ascorbate peroxidase (APX). Besides, MSB up-regulated HMGR (the gene for 3-hydroxy-3-methylglutaryl-CoA reductase) and DXS (the gene for 1-deoxy-D-xylulose-5-phosphate synthase) 24 h after treatment. Both genes are involved in the MVA (mevalonate) and MEP (2C-methylerythritol 4-phosphate) pathways, respectively. These pathways represent the initiation of artemisinin biosynthesis. Thus, MSB application and the consequent up-regulation of early biosynthetic pathway genes along with the triggered oxidative stress may have been contributed to the observed increase in artemisinin production.
García-García, Ana L.; Rodríguez-Ramos, Ruth; Borges, Andrés A.; Boto, Alicia; Jiménez-Arias, David.
Biostimulant activity of Galaxaura rugosa seaweed extracts against water deficit stress in tomato seedlings involves activation of ABA signaling
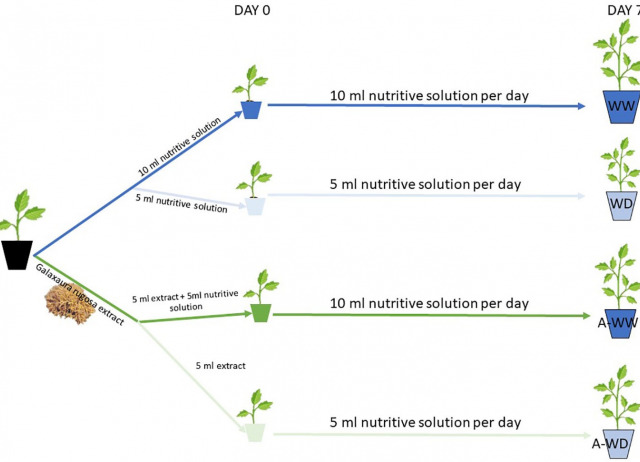
Water scarcity is a serious constraint for agriculture, and global warming and climate change can exacerbate it in many areas. Therefore, sustainable approaches must be implemented to deal with current and future water scarcity scenarios. Genetic and chemical approaches are being applied to manage this limitation and maintain crop yields. In particular, biostimulants obtained from natural sources such as marine algae are promising aids for coping with water deficit stress in agriculture. Here we present a bioprospection study of extracts of the macroalgae Bonnemaisonia hamifera, Galaxaura rugosa, Dasycladus vermicularis, Ulva clathrata, Cystoseira foeniculacea, Cystoseira humilis, Lobophora dagamae, Colpomenia sinuosa and Halopteris scoparia from the north coast of Tenerife, in the Canary Islands. The aqueous extracts of Bonnemaisonia hamifera, Galaxaura rugosa, Dasycladus vermicularis and Cystoseira humilis show biostimulant activity against water deficit stress in tomato seedlings under controlled conditions, providing higher tolerance than the mock-treated control. The Galaxaura rugosa extract showed the highest biostimulant activity against water deficit stress. We demonstrate that this positive effect involves the activation of the abscisic acid (ABA) pathway in Arabidopsis thaliana (arabidopsis) and Solanum lycopersicum (tomato). Application of G. rugosa extract to the root system by drenching tomato seedlings subjected to water deficit leads to improved CO2 assimilation and water use efficiency (WUEp), compared to mock-treated plants. These results highlight a new potential seaweed source of substances with osmoprotectant properties, useful for biostimulant development. Future studies may provide further insight into which components of the seaweed extract induce activation of the ABA pathway.
Morales-Sierra, Sarai; Cristo Luis, Juan; Jiménez-Arias, David; Rancel-Rodríguez, Nereida M.; Coego, Alberto; Rodriguez, Pedro L.; Cueto, Mercedes; Borges, Andrés A.
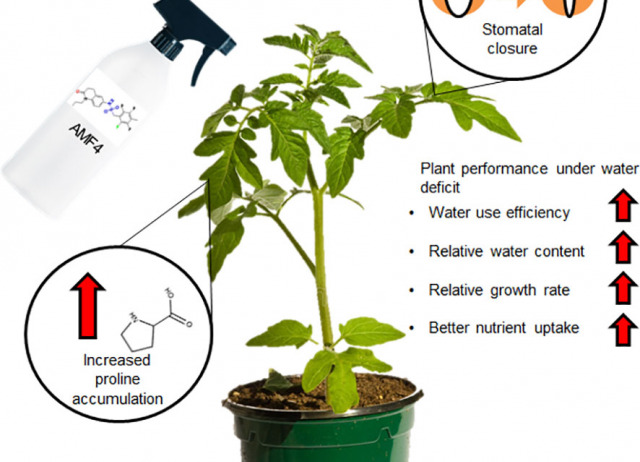
Abscisic acid mimic-fluorine derivative 4 alleviates water deficit stress by regulating ABA-responsive genes, proline accumulation, CO2 assimilation, water use efficiency and better nutrient uptake in tomato plants
Water deficit represents a serious limitation for agriculture and both genetic and chemical approaches are being used to cope with this stress and maintain plant yield. Next-generation agrochemicals that control stomatal aperture are promising for controlling water use efficiency. For example, chemical control of abscisic acid (ABA) signaling through ABA-receptor agonists is a powerful method to activate plant adaptation to water deficit. Such agonists are molecules able to bind and activate ABA receptors and, although their development has experienced significant advances in the last decade, few translational studies have been performed in crops. Here, we describe protection by the ABA mimic-fluorine derivative 4 (AMF4) agonist of the vegetative growth in tomato plants subjected to water restriction. Photosynthesis in mock-treated plants is markedly impaired under water deficit conditions, whereas AMF4 treatment notably improves CO2 assimilation, the relative plant water content and growth. As expected for an antitranspirant molecule, AMF4 treatment diminishes stomatal conductance and transpiration in the first phase of the experiment; however, when photosynthesis declines in mock-treated plants as stress persists, higher photosynthetic and transpiration parameters are recorded in agonist-treated plants. Additionally, AMF4 increases proline levels over those achieved in mock-treated plants in response to water deficit. Thus water deficit and AMF4 cooperate to upregulate P5CS1 through both ABA-independent and ABA-dependent pathways, and therefore, higher proline levels are produced Finally, analysis of macronutrients reveals higher levels of Ca, K and Mg in AMF4- compared to mock-treated plants subjected to water deficit. Overall, these physiological analyses reveal a protective effect of AMF4 over photosynthesis under water deficit and enhanced water use efficiency after agonist treatment. In summary, AMF4 treatment is a promising approach for farmers to protect the vegetative growth of tomatoes under water deficit stress.
Jiménez-Arias, David; Morales-Sierra, Sarai; Suárez, Emma; Lozano-Juste, Jorge; Coego, Alberto; Estevez, Juan C.; Borges, Andrés A. ; Rodriguez, Pedro L.
Addressing the contribution of small molecule-based biostimulants to the biofortification of maize in a water restriction scenario
Biostimulants have become an asset for agriculture since they are a greener alternative to traditionally used plant protection products. Also, they have gained the farmers’ acceptance due to their effect on enhancing the plant’s natural defense system against abiotic stresses. Besides commercially available complex products, small molecule-based biostimulants are useful for industry and research. Among them, polyamines (PAs) are well-studied natural compounds that can elicit numerous positive responses in drought-stressed plants. However, the studies are merely focused on the vegetative development of the plant. Therefore, we aimed to evaluate how drenching with putrescine (Put) and spermidine (Spd) modified the maize production and the yield quality parameters. First, a dosage optimization was performed, and then the best PA concentrations were applied by drenching the maize plants grown under well-watered (WW) conditions or water deficit (WD). Different mechanisms of action were observed for Put and Spd regarding maize production, including when both PAs similarly improved the water balance of the plants. The application of Put enhanced the quality and quantity of the yield under WW and Spd under WD. Regarding the nutritional quality of the grains, both PAs increased the carbohydrates content, whereas the contribution to the protein content changed by the interaction between compound and growth conditions. The mineral content of the grains was also greatly affected by the water condition and the PA application, with the most relevant results observed when Spd was applied, ending with flour richer in Zn, Cu, and Ca minerals that are considered important for human health. We showed that the exogenous PA application could be a highly efficient biofortification approach. Our findings open a new exciting use to be studied deep in the biostimulant research.
Hernandiz, Alba E.; Jiménez-Arias, David; Morales-Sierra, Sarai; Borges, Andres A.; De Diego, Nuria.
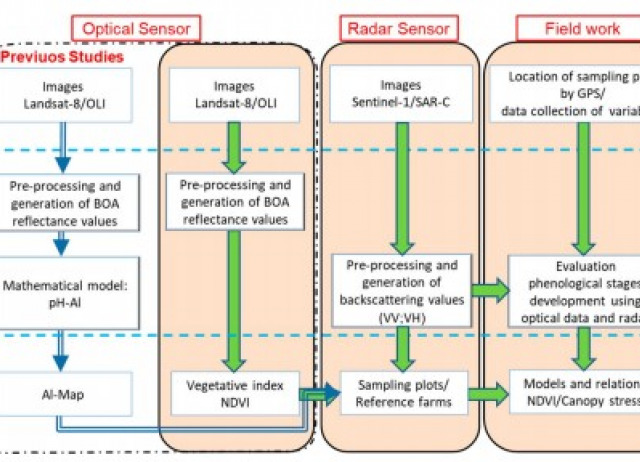
Mapping stressed wheat plants by soil aluminum efect using C‑band SAR images: implications for plant growth and grain quality
Under toxic aluminum (Al) levels in the soil, wheat (Triticum aestivum L.) sufers stress and plant growth is afected. A method for diagnosis of plants is proposed that includes the following as a strategy: to analyze total Al in the soil, employ satellite radar imagery and calculate a vegetation index. The objective of this research, conducted at the feld scale, was to explore how radar backscattering coefcients from a winter wheat canopy, combined with the normalized diference vegetation index (NDVI) and geographic information system (GIS) technology, can be used as a mapping tool for the variability of Al-stressed canopies. As a result, an analysis of covariance showed signifcant diferences, and the lowest plant height was obtained at a high level of soil Al, as well as the minimum grain weight and magnesium content. It was found that a simple model could be used to estimate plant height from the backscattering coefcient of vertical transmit-vertical receive polarization (σ°VV), with a strong correlation (r−0.84). In turn, a third-order polynomial regression model (R20.70) was proposed to estimate the NDVI from σ°VV. This model provided a good estimate of the NDVI at the stem elongation stage of growth (50 days after sowing). Detected NDVI patterns were associated with variation in canopy stress depending on polarimetric information, which, in turn, was related to soil Al levels. Thus, the maps derived from the model can monitor spatial variability, where NDVI values<0.68 indicate stressed areas. This study provides guidance for in-season stress spatial variability caused by Al.
Hernández, Mercedes; Borges, Andrés A.; Francisco‑Bethencourt, Desiderio.
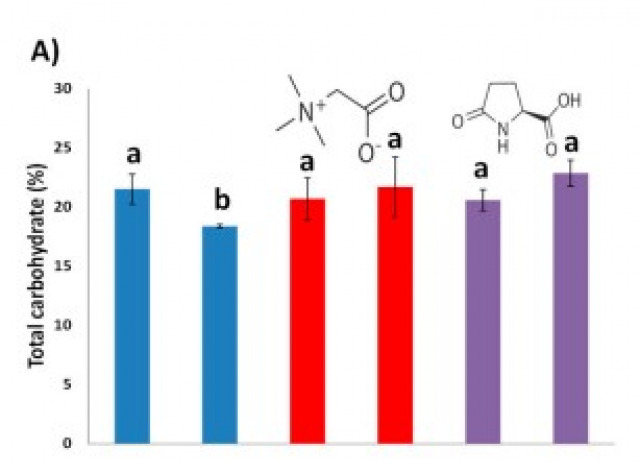
Applying Biostimulants to Combat Water Deficit in Crop Plants: Research and Debate
Climate change has increased the severity of drought episodes by further reducing precipitation in vulnerable zones. Drought induces a substantial decrease in agricultural water, reducing crop yields. Consequently, addressing water consumption can increase farmers’ profits. This work describes lab-to-field research in Zea mays, using two biostimulants: glycine betaine (GB) and Lpyroglutamic acid (PG). The biostimulant optimal dosages were selected using a hydroponic system with 20% polyethylene glycol and nursery experiments under water-deficit irrigation. The established dosages were evaluated in field trials in which irrigation was reduced by 20%. Laboratory biostimulant optimisation showed in stressed treated seedlings (GB 0.1 mM; PG 1 mM) an increased dry weight, relative growth rate and water use efficiency, reducing seedling growth loss between 65 and 85%, respectively. Field trials using a GB-optimised dosage showed an increase in plants’ growth, grain yield and flour Ca content. In addition, grain flour carbohydrate content and protein remained similar to control well-watered plants. Finally, the economic aspects of biostimulant treatments, water consumption, water sources (ground vs. desalinated) and grain biomass were addressed. Overall, GB treatment demonstrated to be a valuable tool to reduce water consumption and improve farmers’ earnings.
Jiménez-Arias, David; Hernándiz, Alba E.; Morales-Sierra, Sarai; García-García, Ana L.; García-Machado, Francisco J.; Luis, Juan C.; Borges, Andrés A.
Root treatment with a vitamin K3 derivative: a promising alternative to synthetic fungicides against Botrytis cinerea in tomato plants
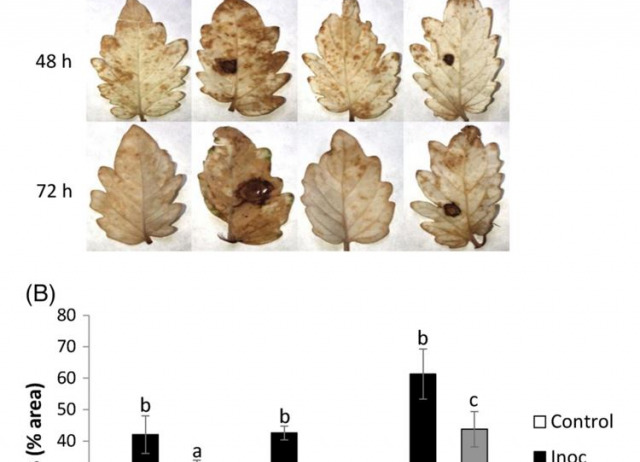
BACKGROUND Botrytis cinerea, the causal agent of gray mold has a great economic impact on several important crops. This necrotrophic fungus causes disease symptoms during vegetative growth and also into postharvest stages. The current method to combat this disease is fungicide application, with high economic costs and environmentally unsustainable impacts. Moreover, there is an increasing general public health concern about these strategies of crop protection. We studied the protection of tomato plants against B. cinerea by previous root treatment with menadione sodium bisulfite (MSB), a known plant defense activator.
RESULTS Root treatment 48 h before inoculation with MSB 0.6 mmol L−1 reduced leaf lesion diameter by 30% and notably cell deaths, compared to control plants 72 h after inoculation. We studied the expression level of several pathogenesis-related (PR) genes from different defense transduction pathways, and found that MSB primes higher PR1 expression against B. cinerea. However, this stronger induced resistance was impaired in transgenic salicylic acid-deficient NahG line. Additionally, in the absence of pathogen challenge, MSB increased tomato plant growth by 28% after 10 days. Our data provide evidence that MSB protects tomato plants against B. cinerea by priming defense responses through the salicylic acid (SA)-dependent signaling pathway and reducing oxidative stress.
CONCLUSION This work confirms the efficacy of MSB as plant defense activator against B. cinerea and presents a novel alternative to combat gray mold in important crops.
García-Machado, Francisco J.; García-García, Ana L.; Borges, Andrés A.; Jiménez-Arias, David